Chronic neuropathic pain can be caused by a broad array of pathologies impacting the central or peripheral nervous systems, such as surgical trauma, spinal cord injury, nerve compression, neurological and infectious diseases, or metabolic and hereditary syndromes. ST-503 is not intended for sporadic or acute pain, but for chronic, intractable pain that completely dominates and often destroys the lives of patients over many years.
SFN has an estimated prevalence of 53 people out of every 100,000 in the U.S., and more broadly, peripheral neuropathies are estimated to affect nearly 40 million Americans. Antidepressants, anticonvulsants, opioids and topical therapies are potential treatment options, although no long-lasting or curative therapies are currently available for SFN patients, leading to a high unmet medical need for this patient population.
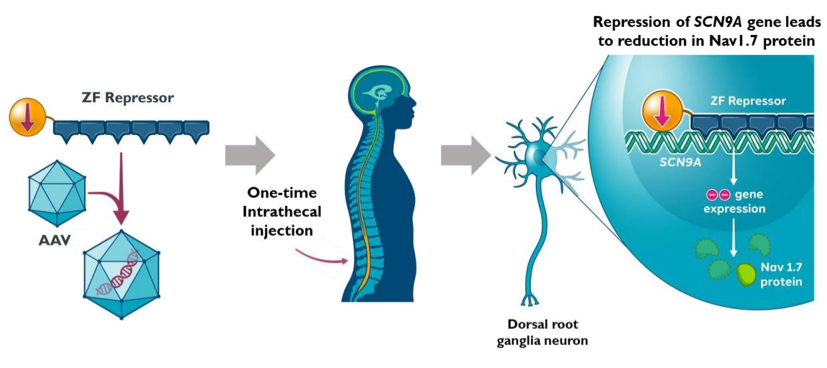
ST-503 uses an AAV vector carrying an engineered zinc finger repressor, or ZFR, to specifically target the human gene, SCN9A, that encodes the Nav1.7 sodium channel and is critical for pain signaling. By directly targeting the SCN9A gene, ST-503 has shown to selectively reduce the expression of Nav1.7 sodium channels in sensory neurons in animal models and significantly reduce pain hypersensitivity, following a single intrathecal administration of ST-503.

Sangamo is recruiting patients in the Phase 1/2 clinical study to assess the safety, tolerability and preliminary efficacy of a one-time dose of ST-503, administered intrathecally to patients with highly debilitating symptoms of burning, prickling, stabbing or “lightning-like” pain due to SFN.
The STAND (Sangamo Therapeutic Analgesic Neuropathic Drug) study is enrolling patients who have had a confirmed diagnosis of SFN-associated refractory pain for at least 6 months.
Food and Drug Administration has cleared the investigational new drug (IND) application for ST-503.