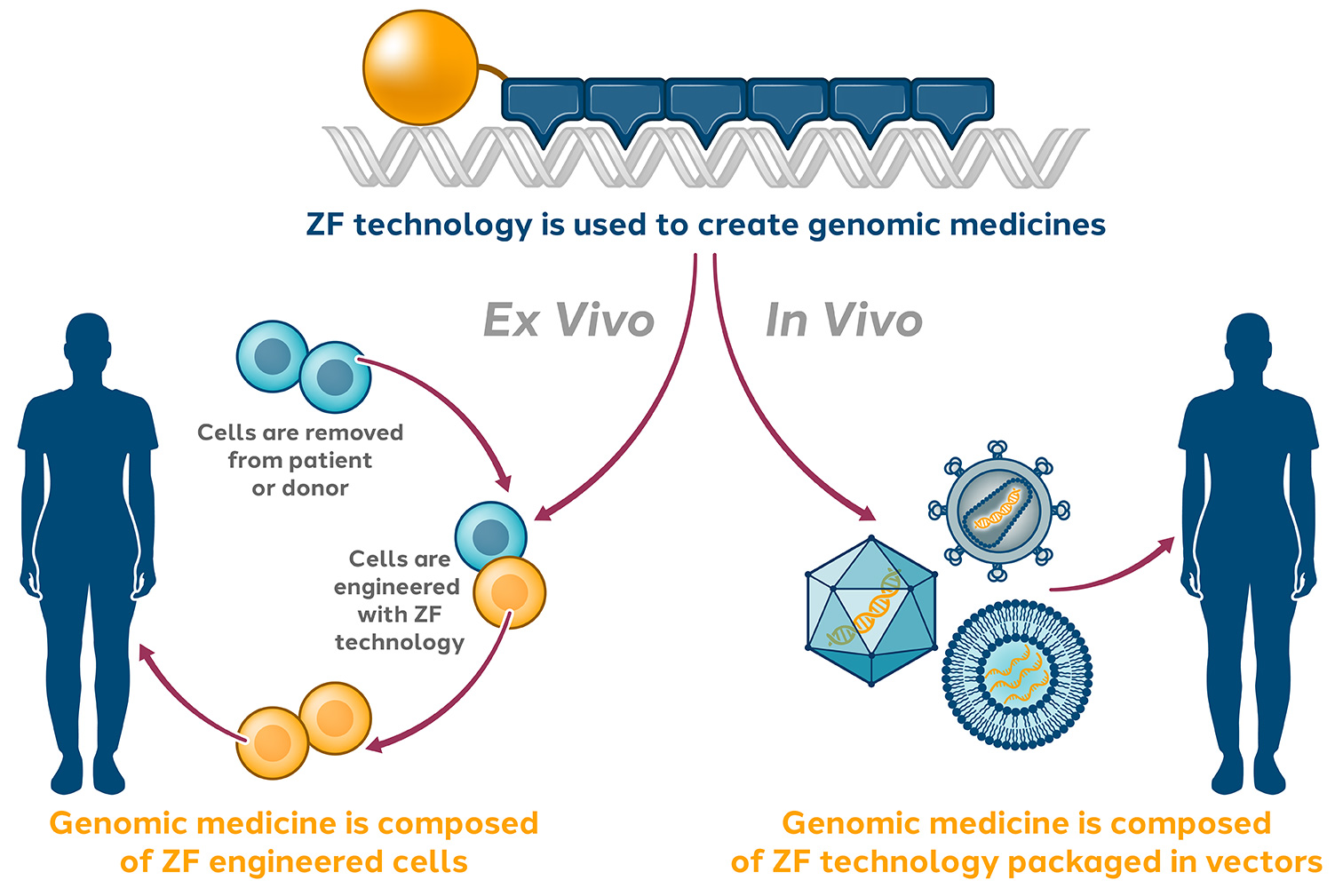
Zinc Finger Platform
Sangamo’s proprietary zinc finger (ZF) platform uniquely confers a number of advantages and has broad therapeutic potential due to its versatility, specificity, clinical utility and compact nature.
Zinc finger technologies are derived from naturally occurring human proteins. They are composed of a DNA-binding domain, which binds with precision to any sequence of DNA, and various functional domains (such as nucleases and transcription factors) based on the therapeutic needs. Due to their compact size, ZF technologies are highly compatible with delivery vectors.
Learn more about our zinc finger platform